Crystal structure models with magnetic features
Generally, our crystal structure models are made as static objects, with no ability to modify or change the structure. Usually, there are good reasons for that - if the real structure doesn't change except under external chemical or physical influence (e.g, oxidation or a phase change), then there is little point in allowing the model structure to change.
But in some materials there are moieties that can be repeatedly removed, replaced and reinserted. Examples are zeolites, which can absorb and release ionic or polar entities such as water, ammonia or metal ions. To model these, we incorporate powerful neodymium magnets into some of the balls to hold the removable parts firmly in place when in the model, but readily removable.

Octahedrally coordinated Mg2+ replaced by H+ in Wadsleyite
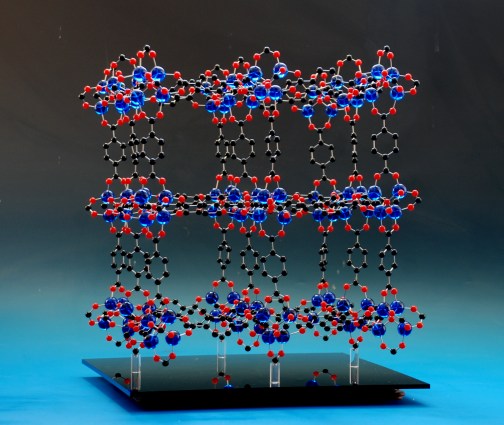
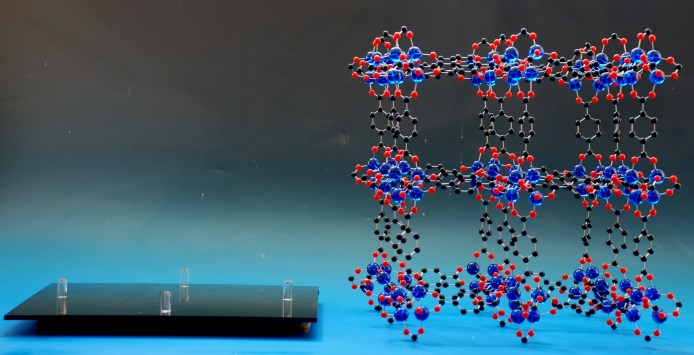
Probably more often, it is not a part of the model that you want to remove, but the entire crystal structure model from a base. By incorporating magnets into the supports in the base, along with corresponding magnets in the atoms in the model, the crystal structure model is held very firmly in place - the model can be held upside down without any danger - but can be readily removed when you want to remove it from its display to show to visitors or students.
Large crystal structure model of a MOF
...and the same model removed from its magnetic base
Company Registered in Scotland no. SC329683 VAT no 916067916